Listed below are the ConcepTests used in class, in the order they appeared. New questions and references to other related questions will be added as they become available. Boldface indicates the correct answer.
These ConcepTests cover Chapters 1-9 in the Brown & Foote text.
ConcepTests for Chapters 10-19 are with the Chem 335 web pages.
ConcepTests for Chapters 20-28 are with the Chem 336 web pages.
Chapter 1 - Bonding & Structure
1. What type of reaction is CH3OH + HCl -----> CH3Cl + H2O
A acid / base
B oxidation / reduction
C addition or elimination
D substitution
2. The Periodic Table has a square for carbon with the numbers
6 and 12.011.
The number 6 represents:
A atomic mass
B atomic weight
C number of protons
D isotope
3. The only structure that does NOT have an error is
D
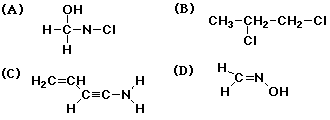
4. The structure below that has an error is D
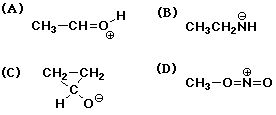
5. Converting between the two resonance forms below (left to right)
involves electron pushing of
A a lone pair from O to C
B a lone pair from C to O
C a pi bond to C
D a pi bond to O
6. The hybridizations of the three carbons in allene (propadiene, CH2=C=CH2) are
A all sp2
B central sp, ends sp2
C central sp3, ends sp2
D some other combination
7. How many carbons are in tryptophane? 11
8. How many hydrogens are in tryptophane? 12
Chapter 2 - Alkanes and Cycloalkanes
For each of the sets of compounds below:
Which compound has the highest boiling point? 9-C, 10-B,
11-B
Which compound has the lowest boiling point? 9-A, 10-A, 11-C
A B C
9. ![]()
10. ![]()
11. ![]()
12, 13. The name for the following compound is
(pick one from each column)
bicyclo
A [6.6] hexane
B [6.6.0] octane
C [4.4.0] decane
D [4.4.2] dodecane
14. The question above requires what level of thinking skills?
A knowledge (1)
B comprehension (2)
C application (3)
D analysis (4)
15. How many isomers have the name
bromomethylcyclopentane? (ignoring chirality)
A 4
B 5
C 6
D 7
16. The rings in each case below can be considered
substituents on the other ring. The orientation
of the rings can be described as:
A cis
B trans
C one cis, one trans
D neither
17.
A equatorial
B axial
C a combination of axial and equatorial
D neither
Chapter 3 - Acid-Base Reactions
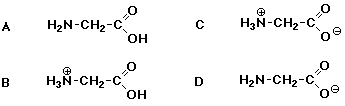
18. The amino acid glycine has two pKa values of 2.35 (carboxylic acid) and 9.78 (protonated amino group). At pH = 7 , glycine looks like: C
Chapter 4 - Stereochemistry
19. How many stereocenters in morphine?
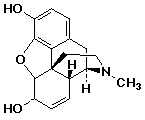
A 4
B 5
C 6
D more than 6
(Note that if N is considered a stereocenter, there are 6)
20. Is this structure (R) or (S) ?
A = R
B = S
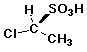
21. Is this structure (R) or (S) ?
A = R
B = S
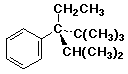
22. The stereochemistry is
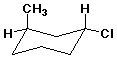
A 1R,3R
B 1R,3S
C 1S,3S
D 1S,3R
23. Pure (S)-2-butanol has a specific rotation
of +13.52°. A sample of 2-butanol prepared
in the lab and purified by distillation has a
calculated specific rotation of +6.76°.
What can you conclude about the composition?
A 50% (S), 50% impurity
B 50% (S), 50% (R)
C 50% (S), 50% racemic
D some other mixture
Chapter 5 - Alkene Structure & Nomenclature
24. All the atoms of ethylene lie in the same plane.
A true
B false
25. All the atoms of allene ( 1,2-propadiene ) lie in one plane.
A true
B false
26. Disubstituted alkenes exhibit cis-trans isomerism.
Do disubstituted allenes exhibit stereoisomerism?
A yes they have cis & trans isomers
B yes they have enantiomers
C yes they have diastereomers
D no
27. How many stereoisomers?
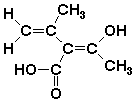
A 2
B 4
C 6
D 8
28. How many stereoisomers?
![]()
A 2
B 4
C 6
D 8
29. How would the bond strength of a C-C single bond compare with the bond strength of a C=C double bond?
A same
B C=C exactly twice C-C
C C=C less than twice C-C
D C=C more than twice C-C
30. How many degrees of unsaturation in benzene?
![]()
A 2
B 3
C 4
D more than 4
31. Is this compound E or Z?
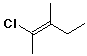
A E
B Z
32. Is it cis or trans?
A cis
B trans
33. Which ONE of the following structures is Z?
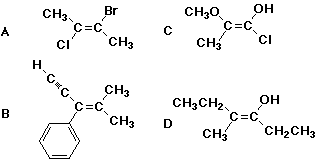
34. Which of the following numberings is correct?
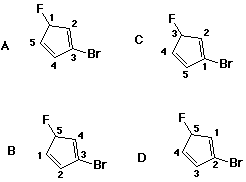 D
D
Chapter 6 - Alkene Reactions
35. Which compound is a possible product from addition of Br2 to 1-butene?
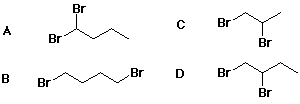 D
D
36. According to Markovnikov's Rule, addition of water to 1-butene should give a
A 1° alcohol
B 2° alcohol
C 3° alcohol
D none of the above
37. If a hypothetical reagent XY gave a syn addition to cyclopentene, the product would be
A cis
B trans
C a mixture of cis and trans
D neither cis nor trans
38. For the potential energy diagram shown, delta H is
A positive (exothermic)
B positive (endothermic)
C negative (exothermic)
D negative (endothermic)
39. For the potential energy diagram shown, Ea is
A positive
B negative
40. Addition of Br2 to 1-butene would give 1,2-dibromobutane which is
A achiral
B racemic
C meso
D optically active
41. Addition of Br2 to cyclopentene would give a product which is
A achiral
B racemic
C meso
D optically active
42. Addition of Br2 to cis-2-butene would give a product which is
A achiral
B racemic
C meso
D optically active
43. Addition of Br2 to trans-2-butene would give a product which is
A achiral
B racemic
C meso
D optically active
44. Addition of OsO4 to cyclopentene would give a product which is
A achiral
B racemic
C meso
D optically active
45. Addition of BH3 followed by H2O2 to trans-2-butene would give a product which is
A achiral
B racemic
C meso
D optically active
46. Addition of BH3 followed by H2O2 to 1,2-dimethylcyclopentene would give a product which is
A achiral
B racemic
C meso
D optically active
47. What are the major products of this reaction, assuming only one equivalent of HCl is available?
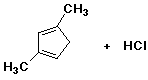
Cl will end up bonded to which positions?
A) 1
B) 2
C) 3
D) a mix of isomers
48. For the potential energy diagram shown, which reaction
is the most exothermic?
A A -> B
B A -> C
49. For the potential energy diagram shown, which reaction
will proceed faster?
A A -> B
B A -> C
50. For the potential energy diagram shown, how would you predict
the ratio of B/C will change when the temperature is increased?
A increase
B decrease
C remain constant
Chapter 7 - Alkyl Halides
51. Which will have the higher boiling point?
A CH3Br
B CH4
52. Which will have the higher boiling point?
A CH3F
B CH4
53. How many unique products are expected from monobromination of isobutane?
A 1
B 2
C 3
D more than 3
54. How many unique products are expected from monobromination of 2-methylbutane?
A 2
B 3
C 4
D more than 4
55. Using the table of bond dissociation energies, calculate delta H for the reaction of cyclohexane with a chlorine atom.
A +3
B -3
C +7
D -7
56. Knowing delta H for the above reaction, you can predict Ea for the reaction will be
A 0
B +7
C -7
D can't tell
57. Chemists actually determine Ea for a reaction by
A measuring product amounts
B measuring rates
C calculating from bond dissociation energies
D calculating from delta H values
Chapter 9 - Alcohols
58. In the reaction shown below, the carbon bonded to Br was originally which carbon?
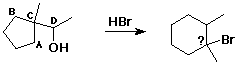
C
59. In the reaction shown below, the six-membered ring is generated by shifting which bond?
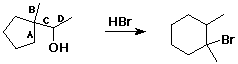
A
60. The pinacol rearrangement of cis-1,2-cyclohexanediol gives which product?
A cyclohexanol
B cyclohexene
C cyclohexanone
D none of the above
61. Periodic acid treatment of cis-1,2-cyclohexanediol gives which product?
A 1,2-cyclohexanedione
B hexanedial
C hexanedioic acid
D none of the above
62. Which of the following does NOT convert an alcohol into a good leaving group for a substitution reaction?
A PBr3
B HBr
C Na
D SOCl2
63. A phase-transfer catalyst is effective because
A it dissolves one of the reagents
B it dissolves both of the reagents
C it dissolves in two immiscible solvents
D it brings an ion into a nonpolar solvent
The next ConcepTests (for Chapters 10-19) are located with the Chem 335 web pages.