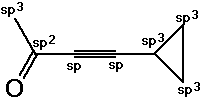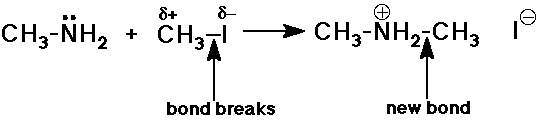Chemistry 331 - Winter 1996
Elements of Organic Chemistry I

Professor Carl C. Wamser

EXAM 1 - Answer Key
February 6, 1996
1. (12 points) Write a complete IUPAC name for each of the following compounds,
including designation of stereochemistry if it is specifically shown:
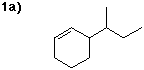
3-sec-butylcyclohexene
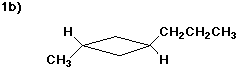
trans-1-methyl-3-propylcyclobutane

(3E)-6-methyl-1,3,5-heptatriene
2. (20 points) Write accurate structures for each of the following names:
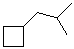
a) isobutylcyclobutane
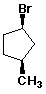
b) cis-1-bromo-3-methylcyclopentane
c) the chair structure for the most stable conformer of tert-butylcyclohexane
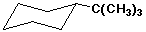
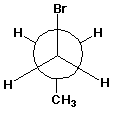
d) a Newman diagram for the most stable conformer of 1-bromopropane
e) (Z)-4,6-dimethyl-3-heptene
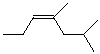
3. (10 points) For the compound shown below:
Write the molecular formula (i.e., CxHy.... ).
C7H15ClO
Identify each carbon atom as 1°, 2°, 3°, or 4°.
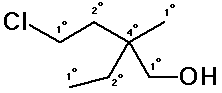
4. (10 points) For the compound shown below:
Write the molecular formula.
C7H8O
Identify the expected hybridization of each carbon atom.
5. (15 points) Predict whether the following reactions would be expected
to have a more favorable equilibrium to the right or to the left.
a) CH3OH + NaNH2 <====> CH3ONa + NH3
pKa = 16 pKa = 33
to the right (CH3OH is the stronger acid)
b) CH3OH + HI <====> CH3I + H2O
delta H = - 13 kcal/mole
Ea = 15 kcal/mole
to the right (the reaction is exothermic)
Write as much as you can of a potential energy diagram for the second
reaction.
Clearly label what the x and y axes represent, the location of the reactants
and the products, and where delta H and Ea would be measured.
Explain what additional information would be needed to make it complete.
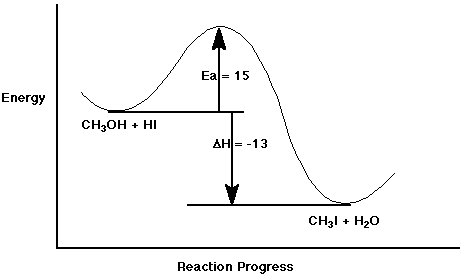
The above potential energy diagram assumes that the reaction occurs
in one step. You would need to know the mechanism in order to know whether
to put in any intermediates.
6. (15 points) The structures shown below are meant to be the two chair
conformations of the same compound. Properly locate the two missing substituents
on the conformation on the right.
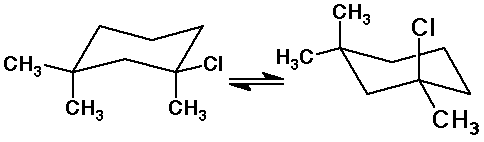
If delta H = - 1.3 kcal/mole for the equilibrium as shown, explain what
information this tells you about the relative interactions between methyl
and chloro substituents.
The equilibrium is favorable to the right.
This indicates that the interaction of an axial Cl with an axial methyl
is not as bad as the interaction of two axial methyls.
7. (18 points) Rewrite each of the reactions shown below, providing more
detailed structures that clearly show the following:
Identify the bonds that are broken.
Identify the new bonds that are made.
Identify which reactant acted as a nucleophile and which as an electrophile.
CH2=O + CH3Li -----> CH3CH2O- Li+

In forming the new C-C bond, CH3 was the nucleophile and CH2 was the
electrophile.
In forming the new O-Li bond, O was the nucleophile and Li was the electrophile.
CH3NH2 + CH3I -----> (CH3)2NH2+ I-
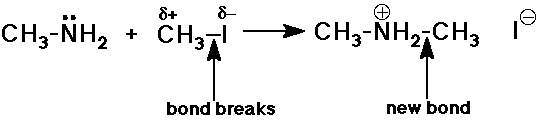
In forming the new C-N bond, N was the nucleophile and CH3 was the electrophile.
Select either of the two reactions and explain why the reactants
have the properties of a nucleophile or electrophile.
See the charge separations shown on the structures above.
![]()
![]()
![]()