![]()
![]()
![]()
1. (15 points) Write a complete IUPAC name for each of the following compounds, including designation of stereochemistry if it is specifically shown:
a) 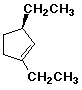
b) 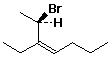
c) 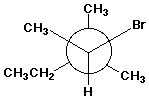
2. (10 points) Examine the pairs of compounds below and identify their relationship to one another. Use the letter codes below:
A - identical structures
B - constitutional isomers
C - conformational isomers
D - diastereomers
E - enantiomers
F - none of the above
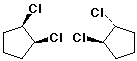 D - diastereomers
D - diastereomers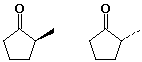 E - enantiomers
E - enantiomers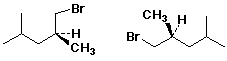 C - conformers
C - conformers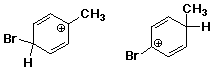 B - constitutional isomers
B - constitutional isomers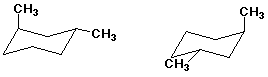 A - identical
A - identical3. (15 points) Complete each of the following reactions by adding the missing part: either the starting compound, the necessary reagents and conditions, or the final major product. Indicate stereochemistry if it is specific.
a) 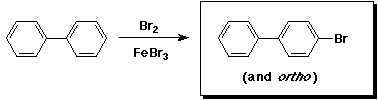
b) 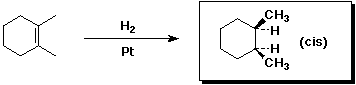
c) 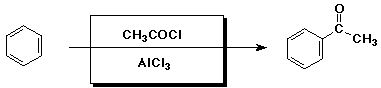
d) 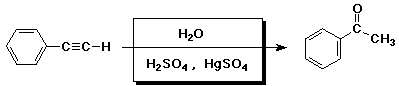
e) 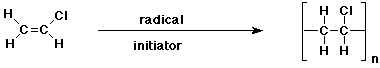
4. (15 points) Write all resonance forms for the cation intermediate that would be formed in the following steps.
a) 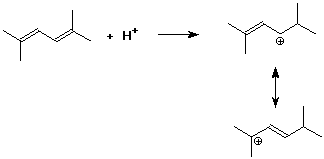
b) 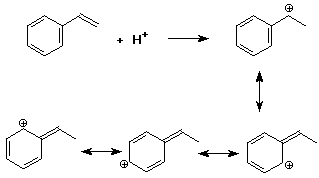
c) 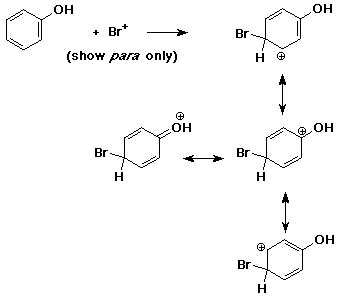
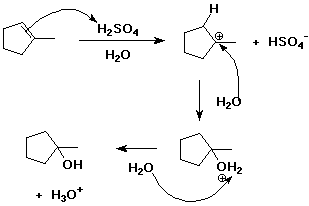
5. (15 points) Write a complete mechanism for the hydration of 1-methylcyclopentene
using aqueous sulfuric acid as catalyst.
Show all steps and use electron-pushing arrows to indicate bonds made and
broken.
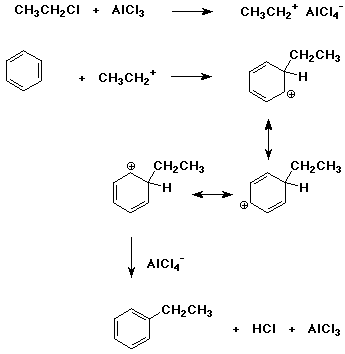
6. (15 points) Write a complete mechanism for the Friedel-Crafts reaction
of chloroethane with benzene, using AlCl3 as catalyst.
Show each step and write all resonance forms for any intermediates involved.
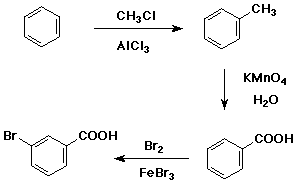
7. (15 points) Write a complete synthetic sequence of reactions that
starts from benzene and creates m-bromobenzoic acid.
Show each reaction step with the necessary reagents and conditions, but
it is not necessary to show reaction mechanisms.