|
|
Organic
Chemistry III
|
|
Professor
Carl C. Wamser
|
|
|
Chapter
20 Notes
|

Carboxyl Derivatives

Carboxyl Derivatives
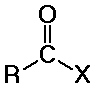
distinguished from aldehydes and ketones because one substituent is NOT
C or H
(usually Cl, O, or N, but it could also be other F, Br, I, S, P, or many other
possibilities)
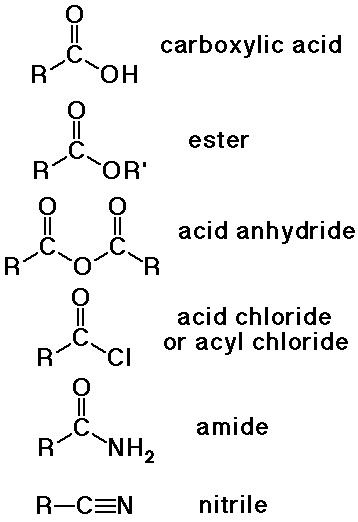
Nomenclature of Carboxyl Derivatives
- acid halides
alkanoyl halide (-oyl halide suffix)
or use common acyl name from acid
- acid anhydrides
alkanoic anhydride (-oic anhydride suffix)
- amides
alkanamide (-amide suffix)
substituents on N use prefix N- (instead of a number location)
- nitriles
alkanenitrile (-nitrile suffix)
- carboxylate salts
metal alkanoate (-oate suffix for anion)
- esters
alkyl alkanoate (a two-part name)
alkyl group is attached to the O
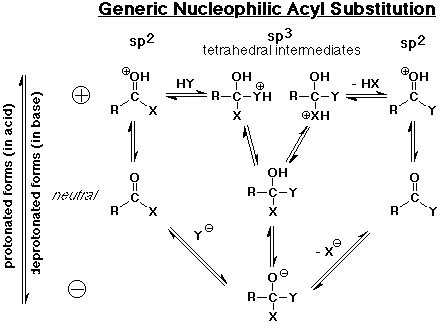
Nucleophilic Acyl Substitution
- the major reaction of carboxyl derivatives
- starts like nucleophilic addition, but becomes substitution when the
leaving group departs
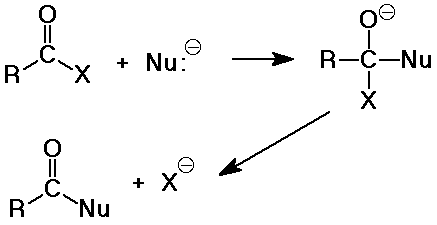
- the tetrahedral intermediate is like that in carbonyl additions
- the product is often another carboxyl derivative
(depnding on what the nucleophile is)
- compared to other nucleophilic substitutions:
SN2 - simultaneous bonding of nucleophile and loss
of leaving group
SN1 - first loss of leaving group, then bonding of
nucleophile (cation intermediate)
acyl - first bonding of nucleophile, then loss of leaving group (anion intermediate)
Reactivity in Acyl Nucleophilic Substitution
- better leaving groups increase reactivity
acid halides > acid anhydrides > esters > amides
- the more reactive derivatives can be easily converted to the others
you can make any of them with the acid chloride - a typical starting point
for synthesis
- all the derivatives can be hydrolyzed to the carboxylic acid
(water as the nucleophile - using acid or base catalysis)
Examples of Acyl Nucleophilic Substitution
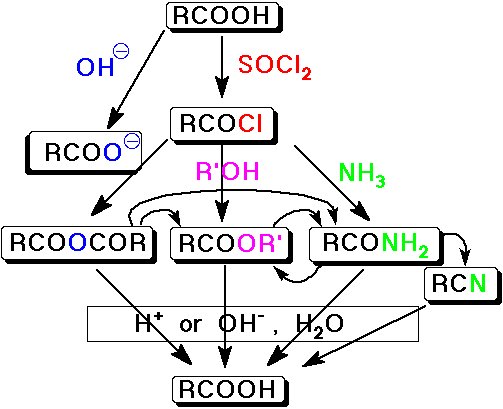
Conversion of Acid Halides into Other Carboxyl Derivatives
- acid halides are the most reactive carboxyl derivative
- they can be converted to all the other derivatives
Conversion of Acid Anhydrides into Other Carboxyl Derivatives
- acid anhydrides are the second most reactive carboxyl derivative
- they can be converted to all the other derivatives except acid halides
but acid chlorides are typically preferred
Synthesis of Esters
- from acid halides plus alcohols
- from acids plus alcohols (Fisher esterification)
- by SN2 reaction of a carboxylate as nucleophile
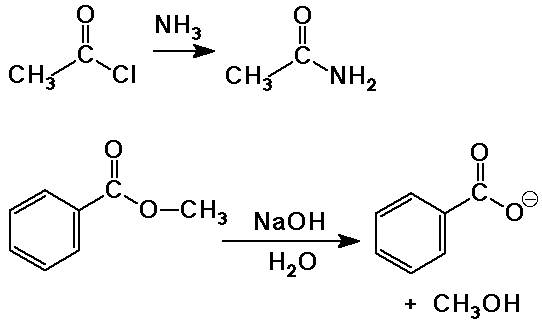
Hydrolysis of Esters
- water as the nucleophile with either acid or base catalysis
- acid-catalyzed hydrolysis is the exact reverse of Fisher esterification
(same mechanism)
- base-catalyzed hydrolysis is often called saponification (soap-making)
saponification is irreversible because a carboxylate salt is formed
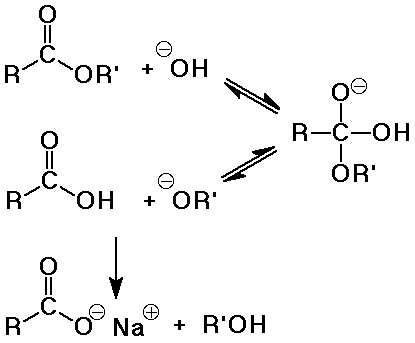
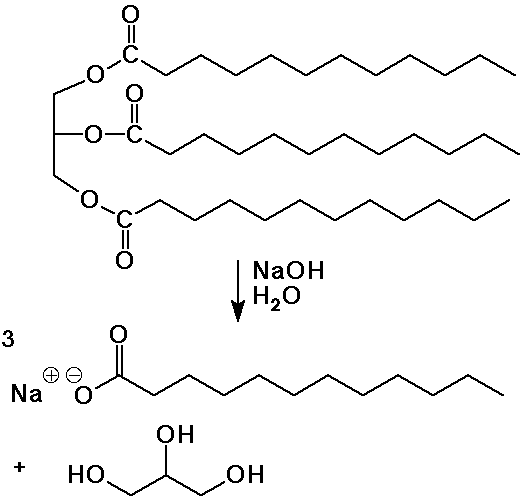
- soaps are the salts of long-chain (fatty) acids ( C8 - C20 )
fats are the esters of fatty acids and glycerol ( 1,2,3-propanetriol )
Other Reactions of Esters
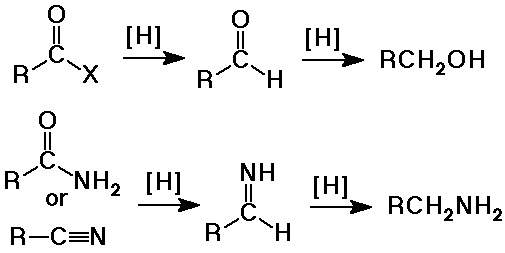
- reduction by LiAlH4 gives primary alcohols (plus
the ester alcohol)
(H- nucleophile first gives substitution - aldehyde intermediate,
then another H- nucleophile gives addition - alcohol product)
- Grignard additions give tertiary alcohols (double addition)
(RMgX nucleophile first gives substitution - ketone intermediate,
then another RMgX nucleophile gives addition - alcohol product)
Synthesis of Amides
- acid chloride plus ammonia (or amine)
Reactions of Amides
- hydrolysis to carboxylic acid - requires prolonged heating
proteins are held together by amide bonds
- reduction by LiAlH4 to amines
Synthesis of Nitriles
- SN2 reaction of primary halides with CN-
- dehydration of amides
Reactions of Nitriles
- nucleophilc additions similar to carbonyl group (polar triple bond)
- hydrolysis to carboxylic acids
- reduction with LiAlH4 to amines
- addition of Grignards to make ketones
Thiol Esters
- Nature often uses sulfur groups as excellent nucleophiles (and leaving
groups)
- coenzyme A contains a thiol group (coA-S-H)
- acetyl CoA acts like acetyl chloride
e.g., acetyl CoA + amines gives acetamides
Summary - Hydride Reductions
Summary - Interconversion of Carboxyl Derivatives

![]()

![]()
![]()
