Organic Chemistry III |
 |
|
Professor Carl C. Wamser |
||
Chem 336 - Spring 2006 |
Exam 2 |
![]()
Organic Chemistry III |
 |
|
Professor Carl C. Wamser |
||
Chem 336 - Spring 2006 |
Exam 2 |
![]()
1. (15 points) Give complete names for each of the following:
a) 
b) 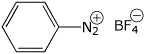
c) 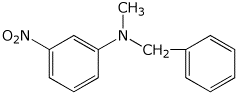
d) 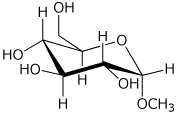
e) 
2. (15 points) Give complete structures for each of the following:
a) the enediol from glyceraldehyde
b) the C-4 epimer of D-glucose in beta-pyranose form
c) alpha-maltose (two D-glucose units joined alpha-1 to 4)
d) the benzyne from p-chlorophenol
e) the Hoffmann elimination product from tetrabutylammonium hydroxide
3. (15 points) Indicate the relative order of the following series of compounds with respect to the property listed. Write “MOST” and “LEAST” under the compounds with the highest and lowest values, respectively.
a) basicity
b) acidity
c) acidity
d) reactivity towards a nucleophile
e) ease of reduction
4. (15 points) Complete each of the following reactions by adding the major parts.
a) 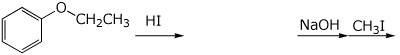
b) 
c) 
d) 
e) 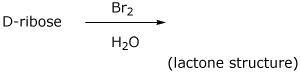
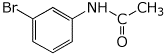
5a. (10 points) Show a synthetic sequence that could be used to prepare m-bromoacetanilide, starting from benzene and acetic acid.
5b. (10 points) In photosynthesis, shown below is the key step in which CO2 is taken in. Number the 5 carbons in the pentose in the standard way, call CO2 carbon 6, and illustrate where the six carbons are located in the next two steps.
Are these balanced reactions?
If not, indicate whether oxidation or reduction is taking place.
6. (20 points) The structure of D-threose is shown below.
a) (3 points) Write an accurate structure for the glycoside that would be formed in the reaction shown below.
b) (3 points) Give a complete IUPAC name for D-threose, including stereochemistry.
c) (4 points) Indicate the products that would be expected on treating D-threose with excess HIO4, and compare the products that would be obtained by treating the glycoside (above) with excess HIO4.
d) (10 points) Write a complete mechanism for the conversion of D-threose to the glycoside shown above using acid catalysis. Show all steps and all resonance forms for any intermediates involved.