 |
|
|
Organic Chemistry III
|
Professor Carl C. Wamser
|

Homework, Chapter 26 Answers

Carey, pages 1103 - 1108 :
Problems 26.16 - 28
1. Olestra, a recent fat substitute, is considered a sucrose polyester,
where the carboxylic acid groups are fatty acids. Write out a possible structure
(or look it up). What aspects of its structure are like a typical fat?
It's considered low-calorie only because it doesn't get digested easily.
If it were digested (completely hydrolyzed), what would the products be?
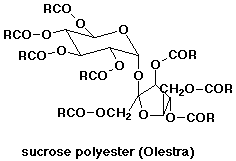
Olestra has a mix of fatty acids attached to the OH groups
of sucrose (typically 6 - 8 fatty acid esters groups). This resembles the
usual triglyceride structure for fats,
so it has a similar "mouth feel", and it behaves similarly in cooking
- even less likely than fats to break down on heating.
If our digestive enzymes could break it down, it would give sucrose (and then
glucose plus fructose) and 6 - 8 fatty acids. Per mole, it would contain more
energy than a typical triglyceride because it has more fatty acids. Per gram,
however, it would be about the same as fats, since the great majority of the
weight in both
molecules is the fatty acid chains.
A concern about Olestra is that it may act as a nonpolar, water-insoluble medium
that absorbs other important nonpolar nutrients (especially Vitamins A and K)
and escorts them out of the digestive tract
rather than allowing them to be absorbed.



![]()
![]()
