Organic Chemistry II |
 |
|
Professor Carl C. Wamser |
||
Chem 335 - Winter 2005 |
Chapter 10 Workshop |
![]()
Organic Chemistry II |
 |
|
Professor Carl C. Wamser |
||
Chem 335 - Winter 2005 |
Chapter 10 Workshop |
![]()
![]()
1. Give the products of the following reactions:
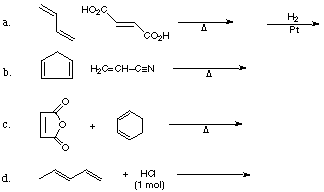
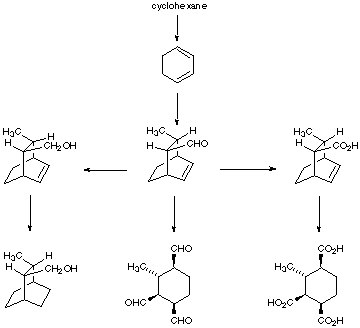
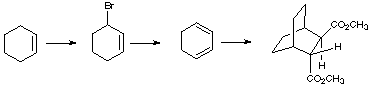
2. Give the reagents or products as appropriate for the following synthetic sequences:
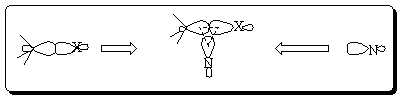
a. Place the relevant molecular orbital(s) (MOs) of the C-X sigma bond on the left side of the paper and the orbital of the nucleophile on the right. Construct the molecular orbitals for the above interaction in between. The nodal properties (i.e., symmetry) and the relative energies of all of the orbitals should be clearly illustrated.
b. Determine whether this interaction of a nucleophile and C-X bond is energetically
favorable or not. What is your prediction about the feasibility of this reaction?
c. Consider now the reaction between an electrophile and the C-X bond using your diagram. Determine if this interaction is energetically favorable or not. What is your prediction about the feasibility of this reaction?
Materials adapted from:
Peer-Led Team Learning: Organic Chemistry, 1/e
Jack A. Kampmeier, University of Rochester
Pratibha Varma-Nelson, St. Xavier University
Donald Wedegaertner, University of the Pacific
Prentice-Hall, 2001, ISBN 0-13-028413-0
http://www.sci.ccny.cuny.edu/~chemwksp/OrganicChem.html