Organic Chemistry II
Dr. Carl C. Wamser
Listed below are the ConcepTests used in class, in the order they appeared. New questions and references to other related questions will be added as they become available. Boldface indicates the correct answer. Numbers indicate an estimate of your responses.
ConcepTests for Chapters 1-9 are with the Chem 334 web pages.
1. This is second-term organic chemistry.
When did you take the first term (Chem 334 or equivalent)?
A last term with you, Dr. Wamser
B last term, somewhere else
C last year, including summer
D longer ago than one year
2. The correct name is:
![]()
A 1-bromo-3-pentyne
B 5-bromo-2-pentyne
3. The correct name is:
![]()
A 2-methylhex-2-en-5-yne
B 5-methylhex-4-en-1-yne
4. The correct name is:
![]()
A hept-2-en-5-yne
B hept-5-en-2-yne
What else does the above name need?
6. Based on the location of alkynes
in the table of acidities (Appendix A2),
which of the following bases would be strong
enough to create an acetylide anion from an alkyne?
A NH3
B CH2=CH2
C NaOH
D CH3Li
7. The correct name is
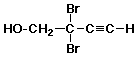
A 2,2-dibromobut-3-ynol
B 2,2-dibromobut-3-yn-1-ol
C 3,3-dibromobut-1-yn-4-ol
D something else
8. In a synthesis of 3-hexyne, a likely compound that
could be the immediate precursor to 3-hexyne is
A 1-butyne
B 1-hexyne
C cis-3-hexene
D cis-3,4-dibromo-3-hexene
9. In a synthesis of 1-butyne, the most likely
immediate precursor to 1-butyne is
A 1-propyne
B 2-butyne
C 1,2-dibromobutane
D bromoethane
10. If the two Br isotopes, 79Br and 81Br,
each has a natural abundance of about 50%,
what is the most likely molecular weight of Br2?
A 158 amu
B 160 amu
C 162 amu
D all three are equally likely
11. A magnetic nucleus with a spin quantum
number of 1 has how many spin state possibilities?
A 1
B 2
C 3
D more than 3
12. The proton NMR spectrum of isobutane consists of
how many absorptions ?
A 1
B 2
C 3
D more than 3
13. The two peaks in the proton NMR spectrum
of isobutane are in what intensity ratio ?
A 1:1
B 2:1
C 3:1
D some other ratio
14. The peak corresponding to the 3° H
in the proton NMR spectrum of isobutane
is split into what pattern ?
A singlet
B doublet
C triplet
D a larger multiplet
15. The peak corresponding to the 1° Hs
in the proton NMR spectrum of isobutane
is split into what pattern ?
A singlet
B doublet
C triplet
D a larger multiplet
16. The compound below would show how many
different absorptions in its 13-C NMR spectrum?
A 2
B 4
C 5
D more than 5
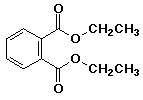
17. The suffix for the IUPAC name for the compound below would be
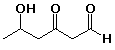
A -ol
B -one
C -al
D -onal
18. The most basic site on an aldehyde is
A C
B O
C H
D the pi bond
19. The compound expected to have the highest boiling point is
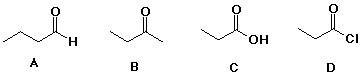
20. Which of the following is a hydrate?
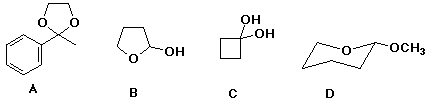
21-23. Which of the above is
an acetal
a ketal
a hemiacetal
Challenge problem: group work (3 - 4 people)
Using only ethylene as a source of carbon, show how to prepare 3-hexanol using Grignard reactions.
Besides using all correct reactions, syntheses are judged on "style" points:
* minimum number of steps
*minimum number of different reagents used
If you were actually carrying out this or a similar synthesis
in lab, what criteria (beyond the number of steps and the number
of reagents) would be involved in deciding whether your synthesis
is a good one?
24. At pH 7, a typical a-aminoacid
will exist primarily in the following form: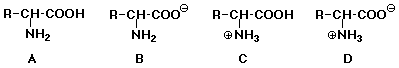
25. Because of the substituent near the carboxylic acid,
an a-aminoacid would be expected to
have a pKa that is
A lower than a typical carboxylic acid
B higher than a typical carboxylic acid
C the same as a typical carboxylic acid
26. Challenge problem - group work (3 - 4 people)
Refer to the table of pKa data on page 627 (#16.29).
a) Explain why the first and second pKa values for a dicarboxylic acid are different from one another.
b) Referring to the pKa of a simple alkanoic acid (pKa ~ 4.8), describe the differences in terms of substituent effects. (instead of charges as your Study Guide does).
c) Explain why the difference is smaller for longer chains.
d) As the chain becomes longer, the limiting values are pKa1 = 4.5 and pKa2 = 5.1 . Why?
Hint - What is the relationship to the pKa of a simple alkanoic acid (pKa ~ 4.8) ?
27. Challenge problem - group work (3 - 4 people)
Hydrolysis of t-butyl acetate can be carried out with either acid or base.
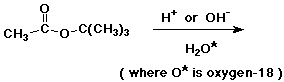
a) In O-18 enriched water, what product or products would be expected to contain O-18 ?
b) In fact, O-18 shows up in different places, depending on whether acid or base is used. Explain.
c) With acid catalysis, isobutylene (2-methylpropene) is a
significant product.
Where does it come from? Why isn't this seen in base?
28. Which is the most acidic hydrogen in this molecule?
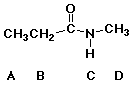
29. Which is the most acidic hydrogen in this molecule?
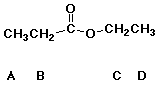
30. Hydrolysis requires only a catalytic amount of acid for each of the following EXCEPT
A esters
B lactones
C amides
D anhydrides
31. The enzyme aldolase catalyzes the reaction of dihydroxyacetone with glyceraldehyde to make D-fructose. This is an aldol reaction. Which carbon acts as the nucleophile?
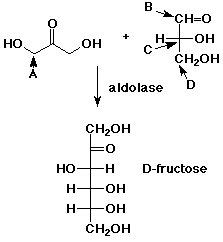
32. Starting with the stereoisomers shown above, how many stereoisomeric products are possible?
A 2
B 3
C 4
D 8
33. If the two starting compounds above reacted in an aldol
reaction in the reverse sense
(the other compound acted as the nucleophile), what product would
result?
34. The Claisen condensation of methyl pentanoate would give which product?
A methyl 5-oxodecanoate
B methyl 3-oxo-2-propylheptanoate
C methyl 2-oxo-2-pentylpentanoate
D some other product
35. After decarboxylation, the above product would give
A 2-decanone
B 5-decanone
C 2-nonanone
D 5-nonanone
36. Which product would NOT be easily made by a malonic ester synthesis?
A octanoic acid
B 2-propylpropanedioic acid
C 2-methylpentanoic acid
D 2,2-dimethylpentanoic acid