Chem 334 - Summer 1998 - Organic Chemistry I
1. (15 pts) Write complete IUPAC names for each of the following compounds, including designation of stereochemistry if it is specifically shown:
a) 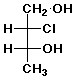
b) ![]()
c) 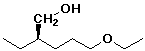
2. (15 pts) Write accurate structures for each of the following:
a) sec-butyllithium
b) DMSO
c) 1,3-dioxacyclopentane
d) the best chair conformation of cyclohexanethiol
e) the transition state for E2 elimination from 2-bromopropane by methoxide
3. (15 pts) The structure shown below is a typical steroid.
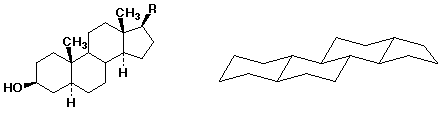
Place the six substituents shown on the structure at left onto the structure at right in their appropriate places (we are considering the two identified H's to be substituents for purposes of this question).
On the structures, identify each of the six substituents as either axial or equatorial.
Explain why each of the ring junctures is called "trans".
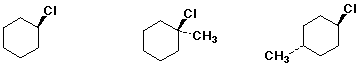
4. (15 pts) Arrange the compounds in each series in order with respect to the property indicated. Write "MOST" and "LEAST" under the appropriate compounds in each group.
a) acidity
![]()
b) radical stability
![]()
c) nucleophilicity
![]()
d) reactivity in an SN2 mechanism
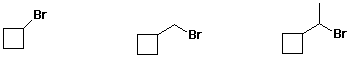
e) amount of axial Cl at equilibrium
5. (15 pts) Complete the following reactions by adding the missing part: either the starting compound, the necessary reagents and conditions, or the final product(s). Indicate stereochemistry when it might be expected to be specific.
a) ![]()
b) ![]()
c) 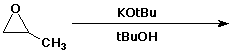
d) ![]()
e) 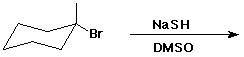
6. (15 pts) Complete the following reactions by adding the missing part: either the starting compound, the necessary reagents and conditions, or the final product(s). Indicate stereochemistry when it might be expected to be specific.
a) 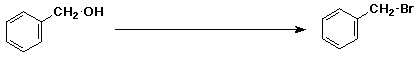
b) 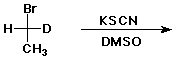
c) ![]()
d) ![]()
e) 
7. (15 pts) Devise a synthetic sequence for either one of the two molecules below. Allowable starting compounds include any alcohol having four or fewer carbons, ethylene oxide (oxacyclopropane), plus any needed solvents or inorganic reagents.
8. (15 pts) Use the table of bond dissociation energies to calculate delta H for each of the following reactions. If the exact data are not available, select the most appropriate analog.
a) hydrolysis of 1-bromopropane
b) hydrolysis of 2-bromopropane
c) From the delta H values calculated above, can you tell which isomer is more reactive? Explain.
9. (16 pts) Write a complete mechanism for the bromination reaction shown below. Show all steps.
(2 pts) If the reaction above began with the (R) enantiomer, the product would be expected to be (circle one):
(R) (S) racemic unknown other
(2 pts) If the reaction above began with the (+) enantiomer, the product would be expected to be (circle one):
(+) (-) (±) unknown other
10. (20 pts) Write complete mechanisms for the following reactions.
Show all steps.
a) ![]()
b) ![]()
11. (15 pts) Write structures for all the different isomers that would have the name 2,3-dimethylcyclopropanol.
Identify enantiomer pairs and meso compounds, but it is not necessary to assign (R) and (S) to the stereocenters (unless you find it helpful).
12. (15 pts) The reaction below has been used as evidence that the shift of a methyl group to a primary carbon is concerted and anti to the leaving group.
(D represents heavy hydrogen.)
Write out the complete mechanism, and explain why this is considered important information about the methyl shift.
13. (10 pts) In order to determine how your body oxidizes ethanol, the two stereospecifically labeled forms of ethanol (shown below) were prepared and subjected to enzymatic oxidation. (D represents heavy hydrogen.)
NAD+ is a cofactor, specifically an oxidant, often used in conjunction with enzymes -- you do not need to know its structure to answer this question.
Explain the significance of the following experimental results.