1. (2 points each) Complete each of the following reactions by adding the missing part: either the starting compound, the necessary reagents and conditions, or the final major product. Indicate stereochemistry if it is specific.
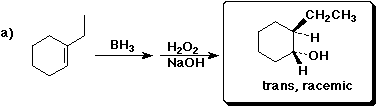
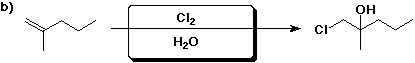
2. (3 points) The reaction shown above in 1a) involves both regioselectivity and stereoselectivity. Write a detailed transition state for the initial addition that explains the reasons for the regiochemistry and stereochemistry observed in this reaction.
The stereochemistry is syn because both the B and H add simultaneously from the same side.
The regiochemistry is "anti-Markovnikov" because the electrophile is the boron, not the hydrogen. The positive charge mainly builds up on the more substituted carbon.
3. (1 point) The product shown above in 1b) is chiral. Describe the expected stereochemistry of the product from this reaction.
The product should be racemic.
4. (2 points) Write a mechanism that shows how the following reaction takes place.