1. (2 points each) Give a complete IUPAC name for each compound below, including designation of stereochemistry if it is specifically shown.
a) 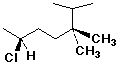
b) 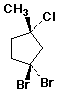
c) 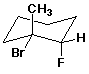
d) 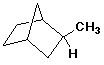
2. (2 points) A new drug is prepared so as to create mainly the (R)(+) enantiomer. The actual synthesis gives a product with an optical purity of 95%. If the specific rotation of optically pure material is +20°, what is the specific rotation of the synthesized sample? What is the composition of the synthesized sample in terms of percentage (R) and (S)?
[a] = + 19°
97.5% (R) + 2.5% (S)