1. (15 points) Write a complete IUPAC name for each of the following compounds, including designation of stereochemistry if it is specifically shown:
a) 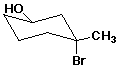
(R,R)-3-bromo-3-methylcyclohexanol
b) 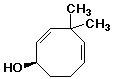
(R)-4,4-dimethyl-2,5-cyclooctadienol
c) 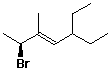
(E,S)-2-bromo-5-ethyl-3-methyl-3-heptene
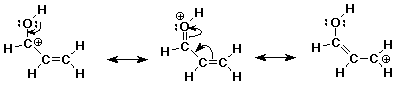
2. (10 points) Add all the necessary lone pairs to the following structure, then show clear electron-pushing arrows to illustrate how to get to all other resonance forms.
3. (10 points) Write the molecular formula and identify the hybridization of all carbons for the compound below.
C7 H6 O
left to right: two sp carbons, two sp3 carbons, three sp2 carbons
4. (10 points) Identify the isoprene units in the compound shown below, specifically indicating heads and tails. Start with the dimethyl location as the first tail.
5. (20 points) Complete each of the following acid base reactions and identify the preferred direction of the equilibrium (right or left).
6. (15 points) Arrange the following in order with respect to the property indicated. Write "MOST" under the most reactive or highest value and "LEAST" under the least reactive or lowest value.
a) nucleophilicity
MID / / MOST / / LEAST
b) basicity
MID / / LEAST / / MOST
c) acidity
MOST / / MID / / LEAST
d) carbocation stability
MOST / / MID / / LEAST
e) rate of solvolysis in methanol
MID / / MOST / / LEAST
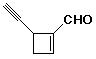
7. (15 points) Complete each of the following reactions by adding the missing part: either the starting compound, the necessary reagents and conditions, or the final major product. Indicate stereochemistry if it is specific.
a) 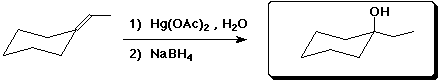
b) 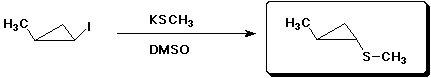
c) 
d) 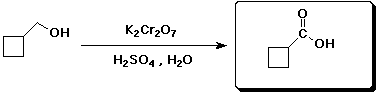
e) 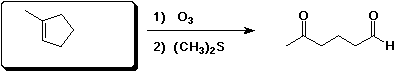
8. (20 points ) Write complete mechanisms for the following reactions. Show all steps and all intermediates involved. Use electron-pushing arrows to indicate electron flow for each step. Show all products of the type indicated.
a) 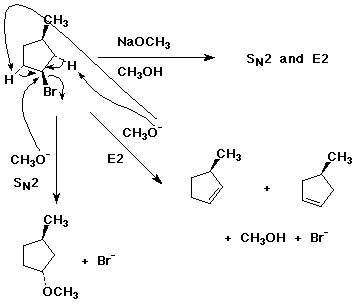
b) 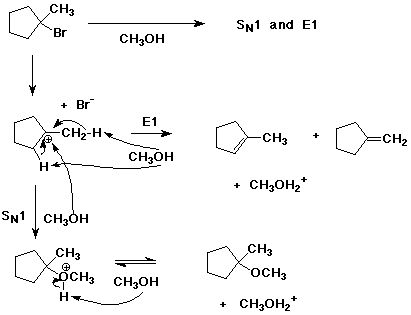
9. (15 points ) Write a complete mechanism for the following reaction. Show all steps and all intermediates involved. Use electron-pushing arrows to indicate electron flow for each step.
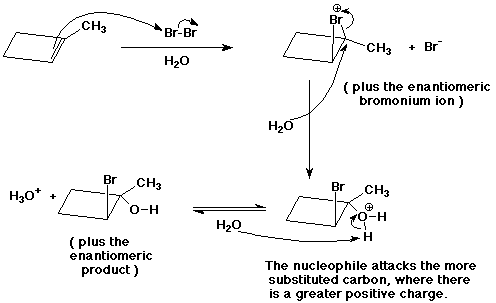
10. (6 points) Refer to the reaction below.
![]()
a) if the starting material is the S enantiomer, the product will be (circle one):
R / / S / / racemic / / can't tell / / other
b) if the starting material has () optical rotation, the product will necessarily be (circle one):
(+) / / () / / (+,) / / can't tell / / other
11. (18 points) Identify the relationship between the following pairs of molecules as one of the following:
A = identical structures, B = constitutional isomers, C = conformational isomers,
D = diastereomers, E = enantiomers, F = resonance structures, G = none of these
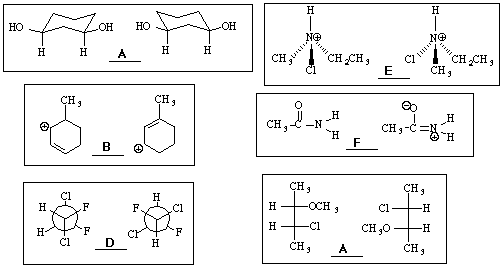
12. (16 points) Use the table of bond dissociation energies to calculate delta H for each of the following reactions.
a) ![]()
delta H = 92 + 103 - 85 - 119 = - 9 kcal/mole
b) ![]()
delta H = 92 + 88 - 70 - 119 = - 9 kcal/mole
c) One of these reactions goes much faster than the other. Predict which one is faster, and explain under what circumstances this is likely to be true.
d) Does the delta H information help you predict which will react faster? Explain why or why not.
13. (30 points) The following synthetic sequence was used to prepare the final compound shown below. Complete all the empty boxes with the missing compounds or reagents and conditions. (3 pts each)
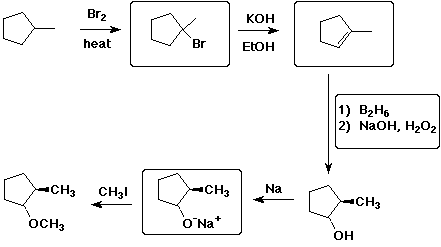
For each one of the five reactions above, describe the reaction
TYPE
(e.g., substitution), and for at least three reactions, describe
the mechanism
(e.g., SN2). (2 pts each)
The final product is optically inactive. Explain why. (2 pts)