Organic Chemistry I |
Exam 2 |
Professor Carl C. Wamser |
![]()
Organic Chemistry I |
Exam 2 |
Professor Carl C. Wamser |
![]()
1. (15 points) Write complete names for each of the following.
a) 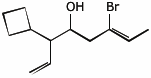
b) 
c) 
2. (15 points) Write accurate structures for the following:
a) the conjugate acid of cyclopentanol
b) the epoxide of 1-methylcyclobutene
c) the polymer of vinylcyclobutane
d) three (3) different primary alcohols of formula C5H12O
e) the transition state of the rate-determining step in the dehydration of t-butyl alcohol
3. (15 points) Arrange the following in order with respect to the property indicated. Write “MOST” under the compound with the highest value and “LEAST” under the compound with the lowest value.
a) stability
![]()
b) heat of hydrogenation (most exothermic = MOST)
![]()
c) reactivity in dehydration
![]()
d) boiling point
![]()
e) reactivity with bromine
![]()
4. (15 points) Complete each of the following reactions by adding the missing part: either the original reactant, the necessary reagents and conditions, or the final major product.
a) ![]()
b) 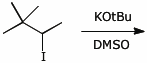
c) ![]()
d) 
e) 
5. (15 points) Use the table of bond dissociation energies to calculate ?H for the reaction below. Use kcal/mol units.
Clearly indicate the bonds broken and the bonds made.
![]()
Write the most likely initiation step and a pair of propagation steps that would result in the overall reaction above. Calculate ?H for each step.
Would this reaction be a reasonable method of preparing alkyl iodides? Explain.
6. (15 points) The reaction shown below follows an SN1 reaction mechanism.
Show all steps to the expected major product, using appropriate electron-pushing arrows.
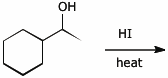
A side reaction also occurs that can be described as an E1 elimination mechanism.
Show all likely elimination products and show the additional steps in the mechanism that would indicate how these products occur.
7. (10 points) Show a sequence of reactions that could be used to accomplish the following conversions.
![]()
![]()
![]()