Organic Chemistry I |
Quiz 7 |
Professor Carl C. Wamser |
![]()
Organic Chemistry I |
Quiz 7 |
Professor Carl C. Wamser |
![]()
1. (3 points) Complete each of the following structures so that they represent the same compound. Add the missing features to the structure on the right so it is the identical compound as the structure on the left.
a) 
b) 
c) 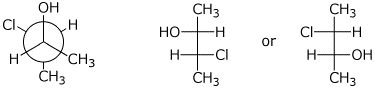
2. (4 points) Give complete names for the following, including designations of absolute configuration.
a) ![]()
(E,R)-hex-3-en-2-ol
b) 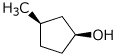
(1S,3R)-3-methylcyclopentanol
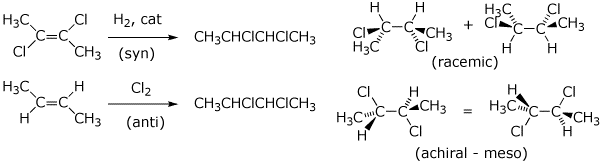
3. (3 points) It’s possible to prepare 2,3-dichlorobutane in two ways, as shown below.
Carefully show the expected stereochemistry of the product(s) in each case.
Indicate whether the products would be expected to be achiral, racemic, or optically active.
![]()