Organic Chemistry I |
Exam 1 |
Professor Carl C. Wamser |
![]()
Organic Chemistry I |
Exam 1 |
Professor Carl C. Wamser |
![]()
1. (16 points) Write complete names for each of the following. Do not include stereochemistry.
a) 
b) 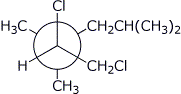
c) 
d) 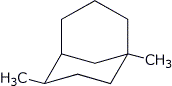
2. (15 points) Write accurate structures for the following:
a) trans-1,3-dimethylcyclobutane

b) the two best resonance forms for CH3-N3 (the three N’s are
linear)
(show all bonds, lone pairs, and formal charges - but accurate 3D is NOT required)

c) the chair conformation of cis-1,3-dichlorocyclohexane that would have the larger dipole moment
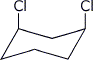
d) a one-carbon compound where the oxidation state of the carbon is +2

e) the chair form of piperidine (shown below), showing the specific location of the lone pair and the H on nitrogen
![]()

3. (15 points) Arrange the following in order with respect to the property indicated. Write MOST and LEAST under the compounds with the highest and lowest values, respectively.
a) acidity
![]()
b) boiling point
![]()
LEAST / / / MOST / / / MIDDLE
c) heat of combustion (amount of heat generated)
![]()
LEAST / / / MIDDLE / / / MOST
d) amount of axial Cl at equilibrium
![]()
LEAST / / / MIDDLE / / / MOST
e) stability of cyclohexane conformation (MOST = most stable)
![]()
LEAST / / / MIDDLE / / / MOST
4. (10 points) Vitamin C is also called ascorbic acid, even though it does not contain the typical carboxylic acid functional group. One (and only one) of the four OH groups is relatively acidic because the conjugate base has resonance forms that delocalize the negative charge to three spots, including two different oxygen atoms. Select the acidic OH group and show the three resonance forms of the conjugate base.
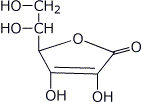

5. (14 points) Complete each of the following acid-base reactions. Identify the acids on each side of the equation, their corresponding pKa values, and predict the preferred direction of the equilibrium.
a) 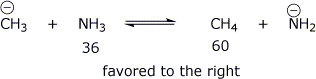
b) 
c) 
Of all the acids and bases in the above three equations,
which is the strongest acid?
H2CO3
which is the strongest base?
CH3-
6. (16 points) Write accurate chair forms for both conformations of each of the following:
a) trans-1-fluoro-4-methylcyclohexane

b) cis-1-fluoro-4-methylcyclohexane
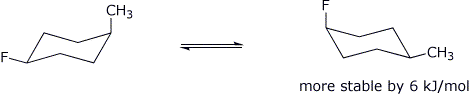
If fluoro prefers equatorial over axial by only 1 kJ/mol, and methyl prefers equatorial over axial by 7 kJ/mol, predict which conformation would be more stable in each case above AND by how much.
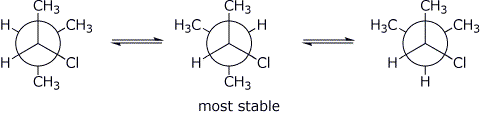
7. (14 points) Write Newman projections for the three staggered conformations of 2-chloro-3-methylbutane, looking down the C2 - C3 bond. Predict the most stable of the three conformations.
![]()