|
|
Organic Chemistry I |
|
Professor Carl C. Wamser |
||
Chapter 7 HW Answers |
![]()
|
|
Organic Chemistry I |
|
Professor Carl C. Wamser |
||
Chapter 7 HW Answers |
![]()
Carey, pages 318 -325 :
Problems 7.25 - 7.48
1. Write complete names for each of the following compounds, including designation of stereochemistry:
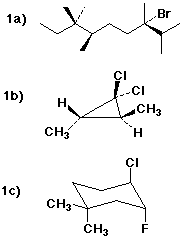
1a) (3S,6R)-3-bromo-2,3,6,7,7-pentamethylnonane
1b) (2S,3S)-1,1-dichloro-2,3-dimethylcyclopropane
1c) (3R,4R)-4-chloro-3-fluoro-1,1-dimethylcyclohexane
2. All of the naturally occuring amino acids are called L because they have a stereochemistry that was historically correlated with one stereoisomer of glyceraldehyde, shown below.

L-Glyceraldehyde and the natural amino acids all have the S absolute configuration. The two exceptions are glycine and cysteine. Look up their 3-dimensional structures and explain why they are different.
Glycine does not have a stereocenter (R
= H).
Cysteine has a sulfur substituent (R = CH2SH), but it does have the same relative configuration as all the other
L-amino acids. Because of the S atom, the priority for the side group is higher
than for the COOH group, which is different than for all the other amino acids
(where the side group priority
falls between the COOH and H).
![]()