|
|
Organic Chemistry I |
|
Professor Carl C. Wamser |
||
Final Exam Answers |
![]()
|
|
Organic Chemistry I |
|
Professor Carl C. Wamser |
||
Final Exam Answers |
![]()
1. (25 points) Write complete names for each of the following, including stereochemistry if it is specifically shown.
a) 
(2R,3S)-3-methyl-1,2,3-pentanetriol
b) 
(4S,5E)-2,6-dimethyl-1,5-octadien-4-ol
c) 
(R)-3-chloro-3-methyl-1,1-cyclohexanediol
d) 
(1R,5R)-5-chloro-3-cycloheptenol
e) 
(R)-1-cyclopentylethanol
2. (15 points) Identify the relationship between the pairs of compounds shown below using the following codes:
I - identical, C - constitutional isomers, D - diastereomers, E - enantiomers,
F - conformational isomers, R - resonance forms, O - other
a)  C
C
b)  I
( or F )
I
( or F )
c)  R
R
d)  C
C
e)  F
F
3. (15 points) Arrange each of the following in order with respect to the property indicated.
Write “MOST” under the compound with the highest value and “LEAST” under the compound with the lowest value.
a) stability
1,4-dimethylcyclohexene / / 4,4-dimethylcyclohexene / / 1,2-dimethylcyclohexene
MIDDLE / / LEAST / / MOST
b) reactivity in an SN1 mechanism
1,2-dimethylcyclohexane / / 1-chloro-2-methylcyclohexane / / 1-bromo-2-methylcyclohexane
LEAST / / MIDDLE / / MOST
c) reactivity in an SN2 mechanism
1-bromo-2,3-dimethylbutane / / 1-bromo-2,2-dimethylbutane / / 2-bromo-2,3-dimethylbutane
MOST / / MIDDLE / / LEAST
d) nucleophilicity
KOH in ethanol / / KOH in DMSO / / ethanol
MIDDLE / / MOST / / LEAST
e) stability
LEAST / / MIDDLE / / MOST
4. (15 points) Complete each of the following reactions by adding the missing component: either the starting compound, the necessary reagents and conditions, or the expected major product. Show stereochemistry if it is specific.
a) 
b) 
c) 
d) 
e) 
5. (25 points) Complete each of the following reactions by adding the expected major product.
Also describe the stereochemistry of the product and describe the mechanism.
a) 
b) 
c) 
d) 
e) 
6. (20 points) Write a sequence of reactions that could be used to prepare the target compounds from the given starting compound.
Show the necessary reagents and conditions for each step and the major compounds formed at each step. Full mechanisms are not needed.
a) ![]()

b) 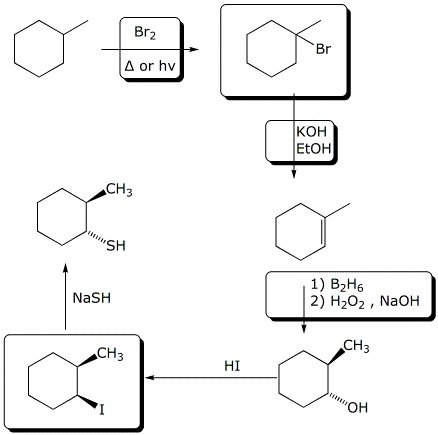
7. (15 points) Using the attached table of bond dissociation energies, calculate ΔH for each of the following reactions.
Use units of kcal/mol. If the exact bond energy is not in the table, use the most appropriate analog you can find.
a) 
b) 
c) 
8. (25 points)
a) (16 points) Complete the following table by entering appropriate numbers
in each blank space (there are 16 spaces to be filled in).
Specific Rotation |
Optical Purity |
Enantiomeric Excess |
% R |
% S |
+ 16° |
100% |
100% |
100% |
0% |
+ 8° |
50% |
50% |
75% |
25% |
0° |
0% |
0% |
50% |
50% |
- 9.6° |
60% |
60% |
20% |
80% |
- 16° |
100% |
100% |
0% |
100% |
b) (9 points) The steroid structure on the left has nine substituents shown. Accurately place each of them on the chair structures at the right in their appropriate locations.

9. (15 points) The free-radical chlorination of 2,2-dimethylbutane gives
three products.
Write names and structures for the three products.
Predict the relative amounts
of each product assuming the relative reactivity of C-H bonds is
3° > 2° > 1° in the ratio 5 : 4 : 1

10. (10 points) Write
a mechanism for the free radical polymerization of propene.
Show initiation
using a peroxide (ROOR) and show at least two propagation steps.

11. (10
points) Write a complete mechanism for the E2 reaction of
cis-1-bromo-2-methylcyclohexane
with KOH in ethanol to form 1-methylcyclohexene.
Use a chair form and electron-pushing arrows to show the stereochemistry
of the mechanism.
12. (10 points) Write a complete mechanism for the solvolysis of tert-butyl chloride in methanol. Show all steps and use electron-pushing arrows.
![]()