![]()
![]()
![]()
McMurry, pp 346-349:
Problems 11.18, 20, 22, 23, 26, 27, 29, 32, 35, 36, 46
1. Condensation reactions seem to have developed more "name" reactions than most other areas of organic chemistry. For each reaction below, identify the alpha-nucleophile and the carbonyl group that would have been involved.
a) Perkin condensation

b) Knoevenagel condensation

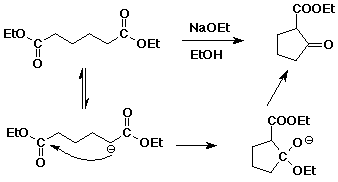
c) Dieckmann condensation
2. A crossed-aldol reaction simply implies that two different components were used, one to act as the nucleophile (electron donor) and one as the carbonyl (electron acceptor). Write a complete mechanism for the crossed aldol reaction between acetaldehyde and benzaldehyde. Indicate which you selected to be the donor and which the acceptor, and also indicate how you might arrange the experiment so that you avoid or minimize the extent of normal aldol reaction.

The nucleophile has to be acetaldehyde, since benzaldehyde doesn't have an alpha-hydrogen and can't make an enolate anion.
To avoid having acetaldehyde react with itself in an aldol reaction, first mix the benzaldehyde with the base (no reaction), then add the acetaldehyde slowly. As the acetaldehyde adds to the reaction mixture, the base will remove a proton to form the enolate anion, which will be surrounded by benzaldehyde, but not much acetaldehyde. The major reaction will be acetaldehyde enolate reacting with benzaldehyde.
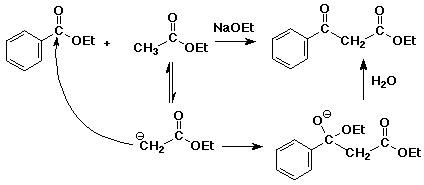
3. Write a complete mechanism for a crossed Claisen condensation between ethyl acetate and ethyl benzoate, using sodium ethoxide as the base.