
Chemistry 332 - Spring 1996
Elements of Organic Chemistry II

Homework, Chapter 9 - Carbonyl Compounds
McMurry, pp 281 - 284:
Problems 9.20, 22-24, 26-32, 34-40, 44, 47
1. Write complete names for each of the following:
a) 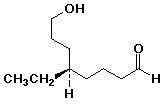
(R)-5-ethyl-8-hydroxyoctanal
b) 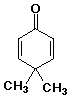
4,4-dimethyl-2,5-cyclohexadienone
2. Write a complete mechanism for the acid-catalyzed formation of the cyclic
acetal between acetone and cis-1,2-cyclopentanediol.
Explain why an acetal is not formed with the trans isomer.

It is sterically difficult for the trans-1,2-diol to get both OH groups
to connect to the same carbon of acetone.
3. Develop a synthetic sequence for 3-ethyl-3-hexanol that uses as organic
starting materials only alcohols having four carbons or fewer.

4. Explain how acetone in the presence of radioactively-labelled water (H2O18)
slowly becomes radioactive itself.

The hydration reaction (addition of water as a nucleophile) does occur,
although the equilibrium constant strongly favors acetone rather than 2,2-propanediol.
Reversal of the reaction could eliminate either normal water or radioactive
water.




