Organic Chemistry III
Dr. Carl C. Wamser
1. (2 points each) Predict the major product from the following reactions:
a) 
b) 
2. (2 pts) Show two different reaction sequences (just the steps with reagents):
one that would convert toluene to p-chlorobenzoic acid
and one that would convert toluene to m-chlorobenzoic acid.

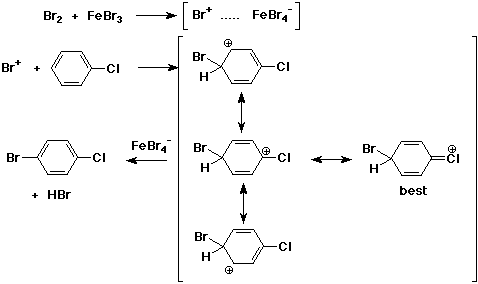
3. (4 pts) Write a complete mechanism for the formation of p-bromochlorobenzene from chlorobenzene. Show all steps and all resonance forms, and identify the most stable resonance form for the intermediate.
Will this bromination occur faster or slower than bromination
of benzene? SLOWER