Welcome to our website
From left to right: Dr. Jenny Zhu, Kathleen Murphy-Strongin, Dr. Ian Munhenzva, Dr. Shawna Vreeke, Megha Gupta, Prof. Robert M. Strongin, Dr. Tendai Mafireyi and Dr. Jiries Meehan-Atrash.
We work in the fields of synthetic and physical organic chemistry, with a focus on redox and chromophore materials and mechanisms. Examples of current studies include the chemistry of electronic cigarettes as well as pharmaceutical, sensor and molecular probe design. The multidisciplinary nature of these topics promotes collaborative research. Local and global collaborations involve the (i) elucidation of reaction pathways and byproducts in electronic cigarettes and hookahs, (ii) targeted imaging of pancreatic cancer, (iii) monitoring of chronic disease, (iv) elucidation of cellular trafficking mechanisms of major pharmaceutical side-effects, and (v) synthesis of highly potent pharmaceuticals that normalize heart arrhythmias via a redox mechanism.
- Wang, L.; Barth, C. W.; Sibrian-Vazquez, M.; Escobedo, J. O.; Lowry, M.; Muschler, J.; Li, H.; Gibbs, S. L.; Strongin, R. M. "Far-red and near-infrared seminaphthofluorophores for targeted pancreatic cancer imaging," ACS Omega, 2017 2, 154-163, doi:10.1021/acsomega.6b00403.
- Jensen, R. Paul; Luo, Wentai; Pankow, James F.; Strongin, Robert M.; Peyton, David H. Hidden Formaldehyde in E-Cigarette Aerosols. N. Engl. J. Med. 2015, 372, 392-394.
- Yang, X.-F.; Huang, Q.; Zhong, Y.; Li, H.; Li, Z.; Lowry, M.; Escobedo,J. O.; Strongin, R. M. A dual emission fluorescent probe enables simultaneous detection of glutathione and cysteine/homocysteine. Chem. Sci., 2014, 5, 2177-2183 (cover feature).
- Sibrian-Vazquez, M., Escobedo, J.O.; Lim, S.; Samoei, G. K.; Strongin, R. M. Homocystamides promote free-radical and oxidative damage to proteins. Proc. Natl. Acad. Sci. USA 2010, 107, 551-554.
- Yang, Y. J.; Lowry, M.; Xu, X. Y.; Escobedo, J. O.; Sibrian-Vazquez, M.; Wong, L.; Schowalter, C. M.; Jensen, T. J.; Fronczek, F. R.; Warner, I. M.; Strongin, R. M. Seminaphthofluorones are a family of water-soluble, low molecular weight, NIR-emitting fluorophores. Proc. Natl. Acad. Sci. USA 2008, 105, 8829-8834.
- Early studies: conjugated materials leading to abiotic biosensors. Our earliest research focused on developing synthetic methods for challenging target molecules with unique redox and/or chromophore properties. Accomplishments included the first synthesis of the fullerene C122, via the reaction of C60 with atomic carbon.
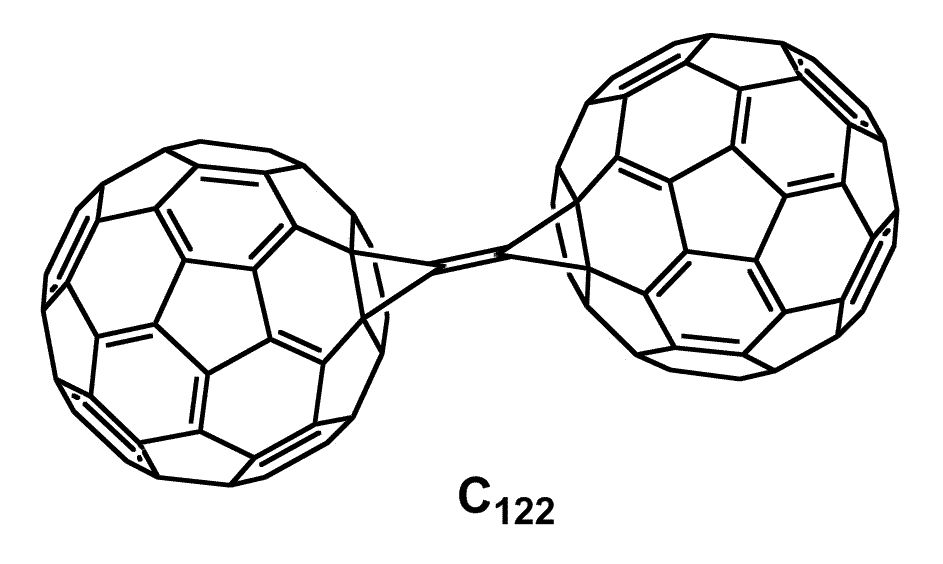 The structure contains two fullerenes linked via a bi(cyclopropylidene) bridge. At the time of the publication, the fullerene-derived dimers, including C121 and C122, and other structurally-related carbon allotrope clusters, had only been observed via mass spectrometry. Our work showed that they could be stable, isolable synthetic targets. This prompted theoretical and experimental interest in analog synthesis as well as in inter-sphere electron transfer effects and materials properties. Meantime, we were investigating a rational synthesis of other unique classes of conjugated materials via a macrocycle based template approach. We developed new synthetic methods for aryl borylation and Suzuki coupling reactions, as well as controlled syntheses of well-defined oligophenylene rods that were successfully coupled to the macrocycle. However, it was troubling to us that solutions of the calixarene-type macrocycles used in these studies developed intense color upon standing, despite the fact that they did not possess conjugation beyond isolated arene rings.
The structure contains two fullerenes linked via a bi(cyclopropylidene) bridge. At the time of the publication, the fullerene-derived dimers, including C121 and C122, and other structurally-related carbon allotrope clusters, had only been observed via mass spectrometry. Our work showed that they could be stable, isolable synthetic targets. This prompted theoretical and experimental interest in analog synthesis as well as in inter-sphere electron transfer effects and materials properties. Meantime, we were investigating a rational synthesis of other unique classes of conjugated materials via a macrocycle based template approach. We developed new synthetic methods for aryl borylation and Suzuki coupling reactions, as well as controlled syntheses of well-defined oligophenylene rods that were successfully coupled to the macrocycle. However, it was troubling to us that solutions of the calixarene-type macrocycles used in these studies developed intense color upon standing, despite the fact that they did not possess conjugation beyond isolated arene rings. 
Although this class of compounds was very well-known, the only prior report on their color formation appeared in a 19th century manuscript by von Baeyer. We eventually determined that trace amounts of xanthene dye chromophores were formed via ring opening and oxidation of the macrocycle. Prompted by the known affinity of arylboronic acids for sugars, we used this material to attain the formation of 12 different solution colors corresponding to 12 different sugar structures, as described in our initial manuscript in the sensor field. At the time, the use of boronic acid dyes for selective colorimetric sugar sensing was still in its infancy. The only prior related example of visual inspection-based selectivity for specific sugars had been reported by Shinkai and co-workers, the most prolific among what was then a limited number of research groups studying related fluorescent sensors.- He, M.; Johnson, R.J.; Escobedo, J.O.; Beck, P.A.; Kim, K.K.; St Luce, N.N.; Davis, C.J.; Lewis, P.T.; Fronczek, F.R.; Melancon, B.J.; Mrse, A.A.; Treleaven, W.D.; Strongin, R.M. Chromophore formation in resorcinarene solutions and the visual detection of mono- and oligosaccharides. J. Am. Chem. Soc. 2002, 124, 5000-5009.
- Willis, D.M. and R.M. Strongin, Palladium-catalyzed borylation of aryldiazonium tetrafluoroborate salts. A new synthesis of arylboronic esters. Tetrahedron Lett. 2000, 41, 8683-8686.
- Davis, C.J.; Lewis, P.T.; McCarroll, M.E.; Read, M.W.; Cueto, R.; Strongin, R.M. Simple and rapid visual sensing of saccharides. Org. Lett. 1999, 1, 331-334 (Highlighted in C & E News).
- Fabre, T.S.; Treleaven, W.D.; McCarley, T.D.l Newton, C.L. Landry, R.M. ; Saraiva, M.C.; Strongin, R.M. The reaction of buckminsterfullerene with diazotetrazole. Synthesis, isolation, and characterization of (C60)2C2. J. Org. Chem. 1998, 63, 3522-3523.
- Simple chemical indicators with high selectivity for specific sugars. Encouraged by the findings summarized above, we tuned the structures of simple organic pH dyes to detect specific bioactive molecules, to help move the field beyond the well-known indicators for protons and metal cations. There was related ongoing research at the time in the boronic acid glucose sensing field.
 We thus focused on creating boronic acid-derived indicators that exhibited selectivity for sugars other than the well-studied glucose or fructose. Towards this end we synthesized the first known boronic acid optical chemosensor that had inherent selectivity for ribose (left, and ribose derivatives) rather than for fructose. This prompted its demonstration as the first indicator for the riboside biomarkers of Adenosyl Lyase Deficiency (ADSL), a devastating inborn error of purine metabolism that is one of the autism spectrum disorders. In related work, we also discovered a chemosensor selective for sialic acid, a sugar acid commonly associated with cancer pathogenesis when expressed at abnormal levels. We extensively investigated and described the mechanistic basis for these examples of previously unreported optical selectivity in the boronic acid sensor field towards expanding the scope and utility of arylboronic acid dyes.
We thus focused on creating boronic acid-derived indicators that exhibited selectivity for sugars other than the well-studied glucose or fructose. Towards this end we synthesized the first known boronic acid optical chemosensor that had inherent selectivity for ribose (left, and ribose derivatives) rather than for fructose. This prompted its demonstration as the first indicator for the riboside biomarkers of Adenosyl Lyase Deficiency (ADSL), a devastating inborn error of purine metabolism that is one of the autism spectrum disorders. In related work, we also discovered a chemosensor selective for sialic acid, a sugar acid commonly associated with cancer pathogenesis when expressed at abnormal levels. We extensively investigated and described the mechanistic basis for these examples of previously unreported optical selectivity in the boronic acid sensor field towards expanding the scope and utility of arylboronic acid dyes.
- Lim, S.; Escobedo, J. O.; Lowry, M.; Strongin, R. M. Detecting specific saccharides via a single indicator. Chem. Comm. 2011, 47, 8295-8297.
- Jiang, S.; Escobedo, J.O.; Kim, K.K.; Alpturk, O.; Samoei, G.K.; Fakayode, S.O.; Warner, I. M.; Rusin, O.; Strongin, R.M. Stereochemical and regiochemical trends in the selective detection of saccharides. J. Am. Chem. Soc. 2006. 128, 12221-12228.
- Yang, Y.J.; Lewis, P.T.; Escobedo, J.O.; St Luce, N.N.; Treleaven, W.D.; Cook, R.L.; Strongin, R. M. Mild colorimetric detection of sialic acid. Collect. Czech. Chem. Comm. 2004, 69, 1282-1291 (invited special issue in honor of Prof. Ivan Stibor).
- Targeted molecular probes and indicators for disease
biomarkers. In order to use selective indicators to address issues
in disease diagnostics beyond sugar detection, we targeted other
bioactive small molecules such as biological thiols, lysophospholipids
and glycolipids.
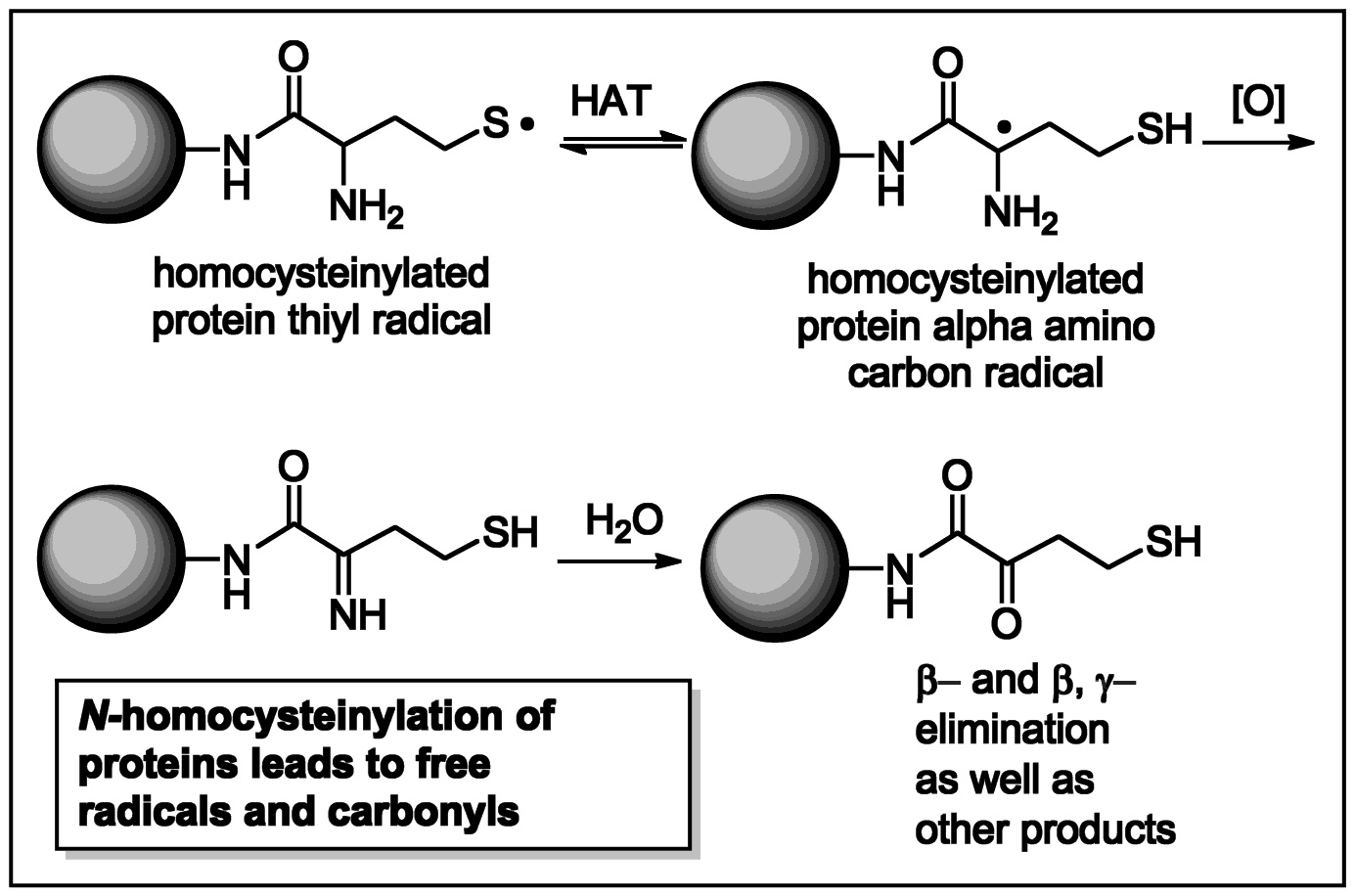 These
studies additionally led to the colorimetric detection of a specific
post-translational protein modification resulting from the reaction of
homocysteine thiolactone with lysine residues. Our manuscript describing
the use of simple aldehyde-bearing xanthene dyes for sensing cysteine
and homocysteine popularized this approach and prompted many related
selective thiol probe syntheses in the field, enabling a variety of
sensing and cellular imaging studies. A relatively more recent report on
the use of acrylate-functionalized dyes for selective biothiol detection
has similarly received significant attention in the community and has
helped initiate many related investigations of acrylate-based "turn-on"
fluorescent indicators. Our project on homocysteine detection led to the
first selective homocysteine colorimetric indicator (no interference
from cysteine or glutathione), via a hydrogen atom transfer mechanism.
These
studies additionally led to the colorimetric detection of a specific
post-translational protein modification resulting from the reaction of
homocysteine thiolactone with lysine residues. Our manuscript describing
the use of simple aldehyde-bearing xanthene dyes for sensing cysteine
and homocysteine popularized this approach and prompted many related
selective thiol probe syntheses in the field, enabling a variety of
sensing and cellular imaging studies. A relatively more recent report on
the use of acrylate-functionalized dyes for selective biothiol detection
has similarly received significant attention in the community and has
helped initiate many related investigations of acrylate-based "turn-on"
fluorescent indicators. Our project on homocysteine detection led to the
first selective homocysteine colorimetric indicator (no interference
from cysteine or glutathione), via a hydrogen atom transfer mechanism.
 The methodology has
enabled homocysteine detection in unprocessed human blood plasma
samples.
The methodology has
enabled homocysteine detection in unprocessed human blood plasma
samples.
In addition, the mechanistic and sensing studies led to the finding that protein homocysteinylation was a source of protein free radicals and carbonyl formation. In other studies, we developed a simple and rapid indicator for glutathione (GSH). It is currently being marketed along with several of our other indicators. In related work, the GSH sensor enabled the development of a convenient blood spot test. We have thus developed simple and useful detection materials and methods for each of the three major biological thiols, cysteine, homocysteine and glutathione.
Many new fluorescent dyes have been synthesized in our group, including the white light emitter (above, looking down into a cuvette) and large Stokes shift dyes that absorb and emit in the near infra-red. These molecules are under study for targeted imaging applications.
- Yang, X. F.; Guo, Y. X.; Strongin, R. M. Conjugate addition/cyclization sequence enables selective and simultaneous fluorescence detection of cysteine and homocysteine. Angew. Chem., Int. Ed. 2011, 50, 10690-10693.
- Sibrian-Vazquez, M., Escobedo, J.O.; Lim, S.; Samoei, G. K.; Strongin, R. M. Homocystamides promote free-radical and oxidative damage to proteins. Proc. Natl. Acad. Sci. USA 2010, 107, 551-554.
- Escobedo, J.O.; Wang, W.H.; Strongin, R.M. Use of a commercially available reagent for the selective detection of homocysteine in plasma. Nat. Protoc. 2006, 1, 2759-2762 (invited protocol manuscript).
- Rusin, O.; St Luce, N.N.; Agbaria, R.A.; Escobedo, J.O.; Jiang, S.; Warner, I.M.; Dawan, F.B.; Lian, K.; Strongin, R.M. Visual detection of cysteine and homocysteine. J. Am. Chem. Soc. 2004, 126, 438-439.
